
However, an exploratory analysis suggested that nivolumab/ipilimumab was more effective than nivolumab alone in the PD-L1 positive group, which was more effective than chemotherapy. “Unfortunately, we did not include a treatment arm with the combination of chemotherapy plus nivolumab in the PD-L1 positive cohort, but only in the negative one,” she said. So it’s a highly manageable treatment.”Ĭonsidering the limitations of the trial, Peters explained that it started before chemotherapy/immunotherapy or immunotherapy alone was approved for the front-line treatment of NSCLC, so the two-drug immunotherapy combination was not compared with current standards of care. Doing this led to a low rate of discontinuation and treatment-related toxicities or deaths. “We used a low dose of ipilimumab (1mg every 6 weeks) to make it tolerable. We can then have an informed discussion with our patients.” She noted that CheckMate-227 showed that safety profiles of both strategies are different, in terms of specific toxicity events encountered, their timing, grade and prevalence, with diarrhoea, rash and fatigue being the most common adverse events with nivolumab/ipilimumab and fatigue, gastrointestinal and haematological toxicities with chemotherapy. The second critical point will be to compare toxicities. The five-year survival from trials with these treatments will teach us if any of the options are better than others. We need to wait for a little more time to see which treatment really gives rise to improved long-term survival. Peters added, “The important step now is to develop an algorithm to select the best front-line treatment for each patient. And now we have a chemotherapy-sparing option of nivolumab plus ipilimumab.” “We already have several front-line treatment options for these patients, including chemotherapy combined with an anti-PD1 agent or an anti-PDL1 agent alone. Solange Peters, Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland. CheckMate-227 is the first trial showing that the combination of nivolumab and ipilimumab prolongs survival as compared to chemotherapy in treatment-naïve patients with metastatic NSCLC,” said study first author Prof. “In my opinion these data are practice changing. Nivolumab, a PD-1 antibody, and ipilimumab, an anti-cytotoxic T-lymphocyte antigen 4 (CTLA-4) antibody, are immune checkpoint inhibitors with distinct but complementary mechanisms of action. The data from the CheckMate-227 trial, reported at the ESMO Congress 2019, suggest that the combination of nivolumab plus low-dose ipilimumab could offer a chemotherapy-free option for first-line treatment of patients with advanced NSCLC. 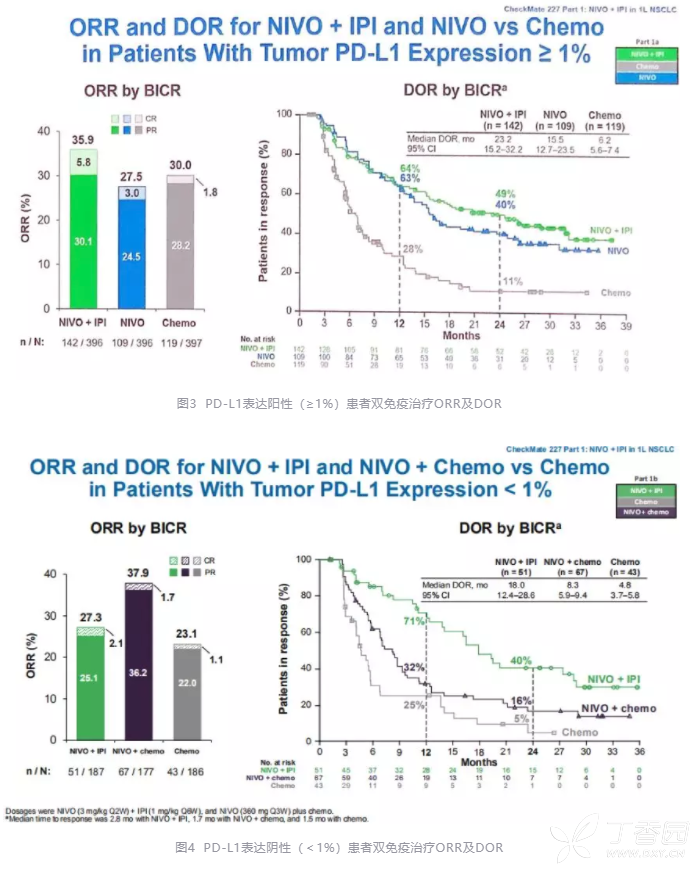
BARCELONA, Spain - New data have shown that first-line treatment with a combination of two immunotherapy drugs improves overall survival in a subset of patients with advanced non-small cell lung cancer (NSCLC) compared to chemotherapy (1).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
